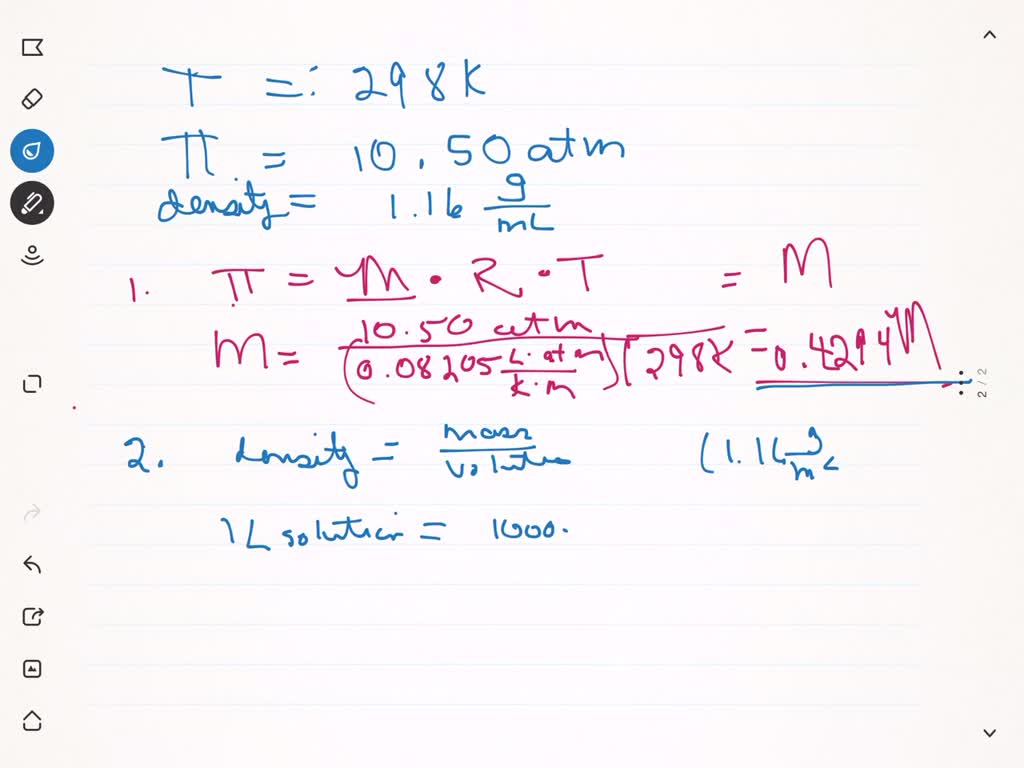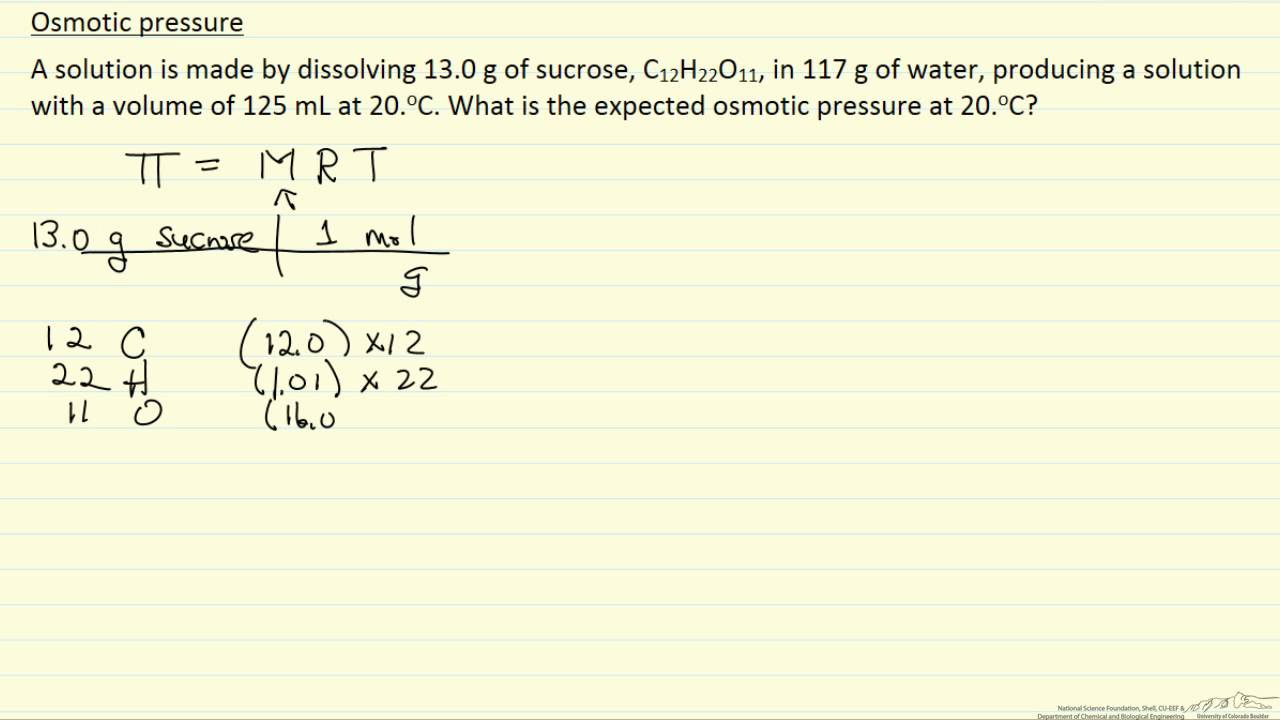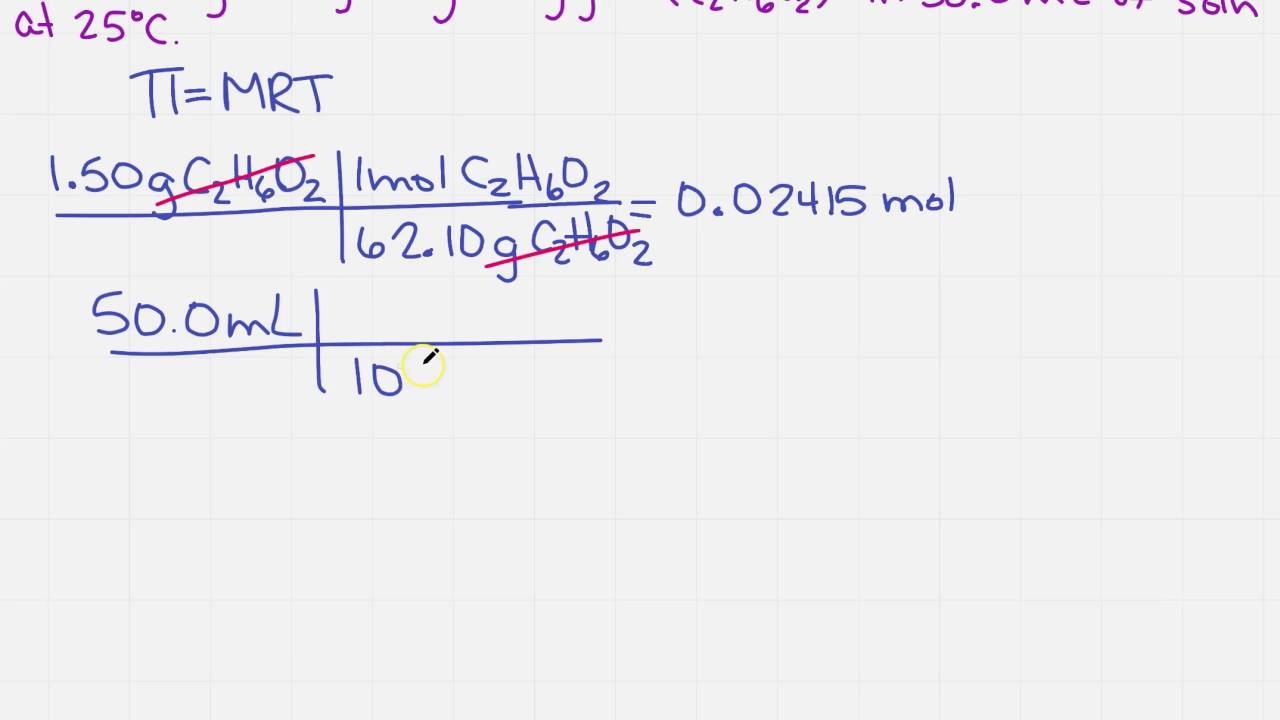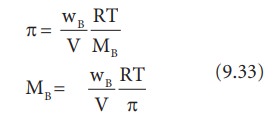![Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ] Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ]](https://i.ytimg.com/vi/PtV0tDX6lCI/maxresdefault.jpg)
Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ]
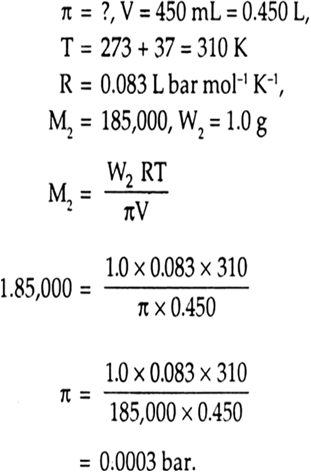
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL at 370C. from Chemistry Solutions Class 12 Haryana Board - English Medium

SOLVED:The osmotic pressure of a 0.010M aqueous solution of CaCl2 is found to be 0.674 atm at 25^∘ C . Calculate the van't Hoff factor, i, for the solution.
Calculate the osmotic pressure of 5% solution of urea at 273K. - Sarthaks eConnect | Largest Online Education Community

C/P osmotic pressure: How do you solve this problem plugging in the numbers to the osmotic pressure formula? : r/Mcat